Report Overview
Preterm Birth And PROM Testing Market Size:
The preterm birth and PROM testing market, with a 6.3% CAGR, is expected to grow to USD 2.521 billion in 2031 from USD 1.748 billion in 2025.
Preterm Birth And PROM Testing Market Trends:
Preterm birth and PROM testing aid in identifying fetal membrane rapture in pregnant women. The patient can be treated and a preterm birth can be avoided by identifying the ruptured fetal membrane. The rising cases of preterm birth and PROM due to several factors such as bad lifestyle, and stress are a major growth driver in the preterm birth and PROM testing market. Moreover, the increasing adolescent pregnancies and preeclampsia cases among pregnant women are further expected to positively impact the preterm birth and PROM testing market. Additionally, government efforts coupled with product launches and research projects by the market players are also contemplated to boost the preterm birth and PROM testing market.
Preterm Birth And PROM Testing Market Growth Drivers:
Rising Cases of Preterm Birth
The increasing number of preterm births due to several factors such as bad lifestyle indicates the widening of the preterm birth and PROM testing market. In 2020, there were reportedly 13.4 million preterm births (births that occurred before 37 full weeks of pregnancy) according to the WHO, and Preterm birth rates in 2020 ranged from 4 to 16% across all nations. Moreover, preterm birth rates are highest in southern Asia and sub-Saharan Africa, and preterm infants in these areas have the highest mortality rates. More than 65% of preterm births worldwide are concentrated in these two areas. The study conducted by the Department of Obstetrics and Gynecology (University of Addis Ababa) in December 2022 revealed that the preterm PROM rate was 2.2% with a perinatal mortality rate of 206/1000.
Adverse Effects of Preterm Birth
There are numerous complications associated with preterm births and PROM which indicates the requirement of early detection and prevention of preterm births thereby propelling the preterm birth and PROM testing market. For instance, the primary factor in the death of children under the age of five is preterm birth problems, which accounted for almost 900,000 deaths in 2019. Breathing issues brought on by an underdeveloped respiratory system, heart issues including patent ductus arteriosus and low blood pressure, and other brief consequences are possible in people born preterm. Bleeding inside the head complications with the digestive system that can lead to necrotizing enterocolitis, anemia, and neonatal jaundice, hypoglycemia, cerebral palsy, learning disabilities, dental troubles, and chronic health conditions like infections, asthma, and feeding difficulties are examples of long-term repercussions that can affect the people born preterm.
Increasing Adolescent Pregnancy and Preeclampsia Cases
Preterm deliveries are closely associated with maternal health hazards, such as adolescent pregnancy and pre-eclampsia. The rising number of adolescent pregnancies and increasing preeclampsia cases are therefore contemplated to augment the preterm birth and PROM testing market. Preeclampsia complications cause 50,000 maternal deaths and 500,000 fetal deaths annually, according to NCBI. The chances of eclampsia, puerperal endometritis, and systemic infections are higher for adolescent mothers (10–19 years old) than for women (20–24 years old), and the risks of low birth weight, preterm birth, and severe neonatal conditions are higher for their offspring. According to the WHO, the anticipated global adolescent birth rate for females aged 10 to 14 in 2022 was 1.5 per 1000 women.
Government Efforts and Research Projects
The government efforts and research projects are encouraging the advancement and adoption of PROM testing thereby propelling the preterm birth and PROM testing market. For instance, in low- and middle-income nations, the WHO conducts research to improve care for pregnant women and preterm infants. These studies include the WHO ACTION Trials (Antenatal Corticosteroids for Improving Outcomes in Preterm Newborns), the Nutritional Management of Growth Flapping in Early Infancy Trial, and an implementation research trial to scale-up immediate kangaroo mother care (KMC). Moreover, the International Federation of Gynecology and Obstetrics (FIGO) launched a special section in the October 2021 issue of the International Journal of Gynecology and Obstetrics. This special section contains nine FIGO good practice recommendations for the effective management and monitoring of preterm birth.
Preterm Birth And PROM Testing Market Geographical Outlook:
North America is expected to grow Significantly
The North American region is expected to hold a significant share of the preterm birth and PROM testing market during the forecast period. Various factors attributed to such a share are the rising rate of preterm births, and technological advancements coupled with increased awareness. For instance, the preterm birth rate increased by 4% in 2021 as compared to 2020 which is the highest since 2007 as per the CDC data, and around one in 10 babies born in the US in 2021 suffered from preterm birth. Moreover, the presence of major market players such as Hologic, Abbott Laboratories, and Sera Prognostics Inc. is further expected to boost the preterm birth and PROM testing market in the region through enhanced accessibility.
List of Top Preterm Birth And PROM Testing Companies:
Hologic Inc. is a US-based multinational company focused on women’s health. Perinatal testing called Rapid fFN® assesses a pregnant woman's risk of having a preterm birth. Other preterm and PROM kits offered by the company are the QuikCheck fFN® Test kit, Rapid fFN® 10Q Cassette Kit.
Sera Prognotics Inc. is one of the leading women’s health companies headquartered in the US. In February 2023, the company announced the positive top-line data from Avert Preterm Trial in the reduction of neonatal morbidity or neonatal death. It was conducted at ChristianaCare in Wilmington.
Preterm Birth And PROM Testing Market Scope:
| Report Metric | Details |
|---|---|
| Study Period | 2021 to 2031 |
| Historical Data | 2021 to 2024 |
| Base Year | 2025 |
| Forecast Period | 2026 – 2031 |
| Companies |
|
Report Metric | Details |
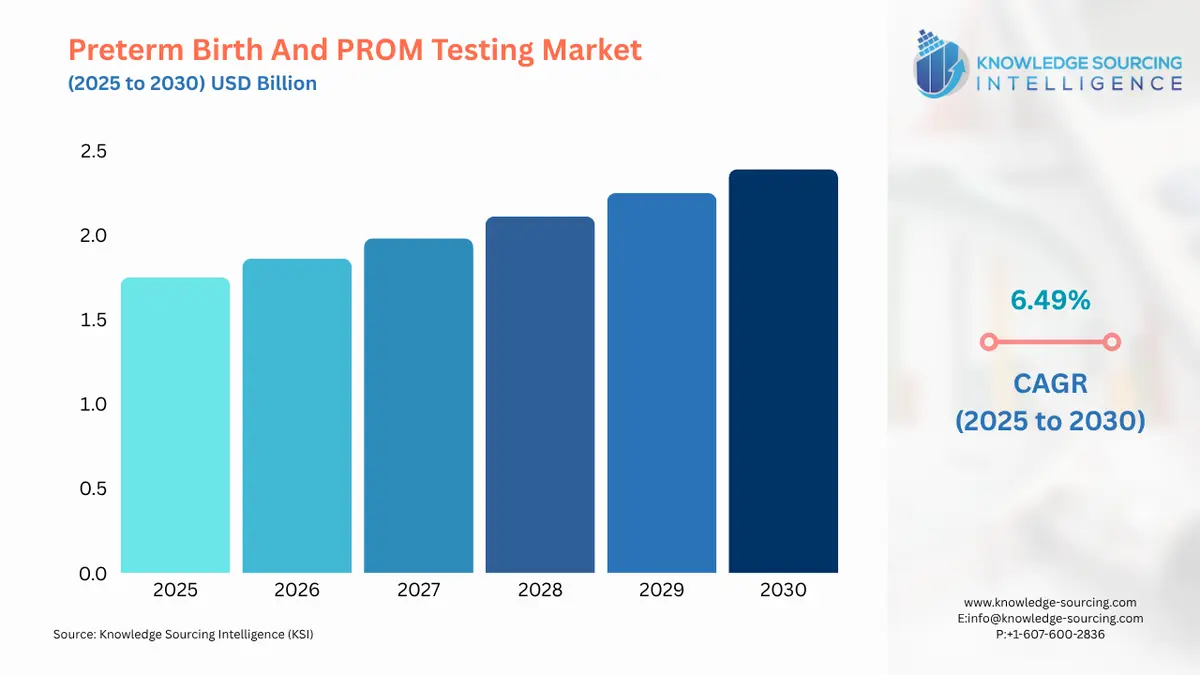
Preterm Birth And PROM Testing Market Size in 2025 | USD 1.748 billion |
Preterm Birth And PROM Testing Market Size in 2030 | USD 2.393 billion |
Growth Rate | CAGR of 6.49% |
Study Period | 2020 to 2030 |
Historical Data | 2020 to 2023 |
Base Year | 2024 |
Forecast Period | 2025 – 2030 |
Forecast Unit (Value) | USD Billion |
Segmentation |
|
Geographical Segmentation | North America, South America, Europe, Middle East and Africa, Asia Pacific |
List of Major Companies in the Preterm Birth And PROM Testing Market |
|
Customization Scope | Free report customization with purchase |
Preterm Birth and PROM Testing Market Segmentation
By Preterm Birth Tests
Pelvic Exam
Ultrasound
Others
By PROM Tests
Nitrazine Test
Ferning Test
Others
By End-User
Diagnostic Laboratories
Hospitals
Others
By Geography
North America
USA
Canada
Mexico
South America
Brazil
Argentina
Others
Europe
Germany
France
United Kingdom
Spain
Others
Middle East and Africa
Saudi Arabia
UAE
Others
Asia Pacific
China
India
Japan
South Korea
Indonesia
Thailand
Others
Our Best-Performing Industry Reports:
Market Segmentation
By Preterm Birth Tests
By Prom Tests
By End-user
By Geography
Table of Contents
1. EXECUTIVE SUMMARY
2. MARKET SNAPSHOT
2.1. Market Overview
2.2. Market Definition
2.3. Scope of the Study
2.4. Market Segmentation
3. BUSINESS LANDSCAPE
3.1. Market Drivers
3.2. Market Restraints
3.3. Market Opportunities
3.4. Porter’s Five Forces Analysis
3.5. Industry Value Chain Analysis
3.6. Policies and Regulations
3.7. Strategic Recommendations
4. TECHNOLOGICAL OUTLOOK
5. PRETERM BIRTH AND PROM TESTING MARKET BY PRETERM BIRTH TESTS
5.1. Introduction
5.2. Pelvic exam
5.3. Ultrasound
5.4. Others
6. PRETERM BIRTH AND PROM TESTING MARKET BY PROM TESTS
6.1. Introduction
6.2. Nitrazine Test
6.3. Ferning Test
6.4. Others
7. PRETERM BIRTH AND PROM TESTING MARKET BY END-USER
7.1. Introduction
7.2. Diagnostic Laboratories
7.3. Hospitals
7.4. Others
8. PRETERM BIRTH AND PROM TESTING MARKET BY GEOGRAPHY
8.1. Introduction
8.2. North America
8.2.1. USA
8.2.2. Canada
8.2.3. Mexico
8.3. South America
8.3.1. Brazil
8.3.2. Argentina
8.3.3. Others
8.4. Europe
8.4.1. Germany
8.4.2. France
8.4.3. United Kingdom
8.4.4. Spain
8.4.5. Others
8.5. Middle East and Africa
8.5.1. Saudi Arabia
8.5.2. UAE
8.5.3. Others
8.6. Asia Pacific
8.6.1. China
8.6.2. India
8.6.3. Japan
8.6.4. South Korea
8.6.5. Indonesia
8.6.6. Thailand
8.6.7. Others
9. COMPETITIVE ENVIRONMENT AND ANALYSIS
9.1. Major Players and Strategy Analysis
9.2. Market Share Analysis
9.3. Mergers, Acquisitions, Agreements, and Collaborations
9.4. Competitive Dashboard
10. COMPANY PROFILES
10.1. Hologic, Inc
10.2. Abbott
10.3. Sera Prognostics Inc.
10.4. CooperSurgical Inc.
10.5. IQ Products
10.6. Qiagen N.V.
10.7. BHR Pharmaceuticals
10.8. NX Prenatal, Inc.
11. APPENDIX
11.1. Currency
11.2. Assumptions
11.3. Base and Forecast Years Timeline
11.4. Key benefits for the stakeholders
11.5. Research Methodology
11.6. Abbreviations
LIST OF FIGURES
LIST OF TABLES
Request Customization
Tell us your specific requirements and we will customize this report for you.
Download Free Sample
Get a sample copy of this report with charts, TOC, and methodology.
Speak to Analyst
Ask our analysts any questions you have about this market research report.
Preterm Birth And PROM Testing Market Report
Trusted by the world's leading organizations











