Report Overview
Human Milk Oligosaccharides (HMO) Milk Powder Market Size:
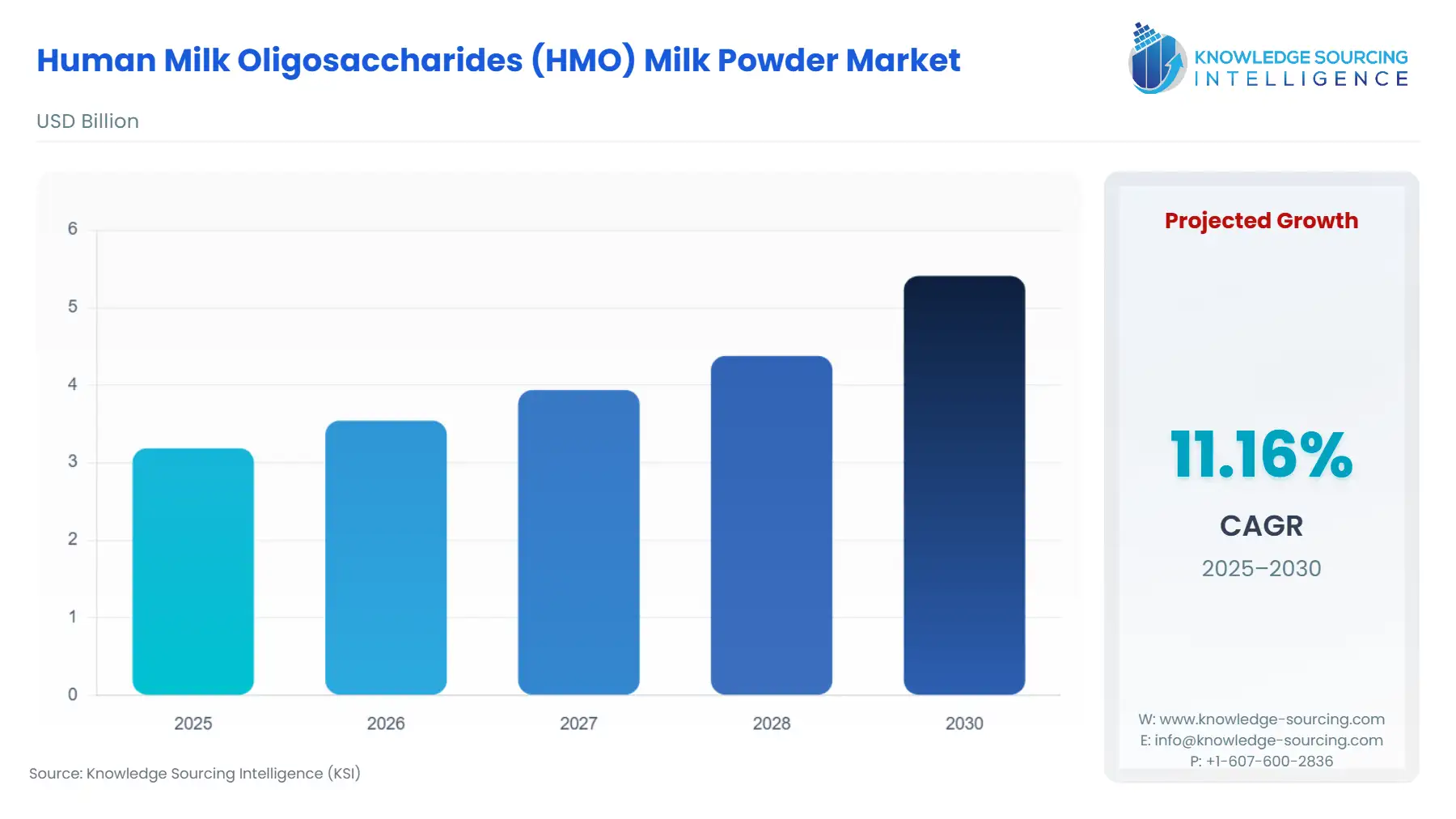
The Human Milk Oligosaccharides (HMO) Milk Powder Market is set to witness robust growth at a CAGR of 11.2% during the forecast period, to be worth USD 5.4 billion in 2030 from USD 3.2 billion in 2025.
Human Milk Oligosaccharides (HMO) Milk Powder Market Introduction:
The Human Milk Oligosaccharides (HMO) Milk Powder Market represents a rapidly evolving segment within the broader pediatric nutrition industry, driven by advancements in biotechnology and a growing emphasis on replicating the functional benefits of human breast milk in infant formula. HMOs are complex carbohydrates naturally present in human breast milk, recognized as the third most abundant solid component after lactose and lipids. These indigestible sugars play a critical role in promoting gut microbiome health, enhancing infant immunity, and supporting cognitive development, making them a cornerstone of premium infant nutrition.
The market for HMO-enriched milk powders is expanding as manufacturers strive to develop breast milk substitutes that closely mimic the nutritional and immunological benefits of breastfeeding, addressing the needs of infants who cannot be breastfed. The HMO milk powder market is a specialized niche within the early life nutrition market, focusing on infant formulas and follow-on milk powders fortified with HMOs to support gut microbiome health and infant immunity. These bioactive compounds, including 2’-Fucosyllactose (2’-FL), Lacto-N-neotetraose (LNnT), and 3’-Sialyllactose (3’-SL), act as prebiotics that foster beneficial gut bacteria like Bifidobacterium, which are essential for digestive health and immune system development. The World Health Organization emphasizes exclusive breastfeeding for the first six months of life, but for infants reliant on formula, HMO-enriched products offer a scientifically backed alternative that narrows the gap between formula and breast milk. The market is witnessing significant innovation, with companies like Nestlé, Abbott, and Chr. Hansen is leading the integration of HMOs into premium infant nutrition products. For example, Nestlé’s 2023 launch of the Wyeth Illuma formula with 2’-FL and LNnT in China marked a milestone in delivering breast milk substitutes tailored to regional nutritional needs. The early life nutrition market is increasingly driven by parental demand for products that support long-term health outcomes, such as reduced infection risks and enhanced cognitive development. HMOs contribute to these outcomes by modulating the gut microbiota, producing short-chain fatty acids, and preventing pathogen adhesion in the infant gut. Recent studies, such as a 2024 clinical trial, demonstrated that HMO-supplemented formulas increase Bifidobacterium levels and improve stool consistency, aligning formula-fed infants’ microbiomes closer to those of breastfed infants. The market also extends beyond infants, with HMOs being explored for adult nutrition in functional foods and supplements, reflecting their versatility in promoting gut microbiome health across age groups.
Several factors drive the market growth, including:
Demand for Premium Infant Nutrition: Parents and healthcare professionals prioritize premium infant nutrition products that replicate the benefits of breast milk, driving demand for HMO-enriched milk powders. This is particularly pronounced in regions with high working-mother populations, where breastfeeding rates tend to be lower.
Advancements in Biotechnology: Innovations in precision fermentation have enabled scalable production of HMOs, making them viable for widespread use in pediatric nutrition. For instance, Chr. Hansen’s 2024 biosafety approval for its MyOli 5-HMO blend in China underscores the role of biotechnology in market growth.
Focus on Gut Microbiome Health: Growing awareness of the gut microbiome’s role in infant immunity and long-term health fuels demand for HMO-based products that support beneficial bacteria and reduce infection risks.
Regulatory Approvals: Recent regulatory milestones, such as China’s 2023 approval of 2’-FL and LNnT as food nutrition enhancers, facilitate market expansion by enabling HMO integration into infant formulas.
The market faces challenges, including:
High Production Costs: The complex processes involved in HMO production, such as precision fermentation, result in high costs, limiting affordability in price-sensitive markets.
Regulatory Complexity: Stringent and varying global regulations for HMO use in pediatric nutrition can delay product launches and increase compliance costs, hindering market growth.
Production of HMOs for Commercial Use:
HMOs are produced commercially primarily through precision fermentation, a biotechnology process that uses genetically engineered microorganisms, such as E. coli or yeast, to synthesize specific oligosaccharides like 2’-FL and LNnT. This method involves inserting genes that encode HMO-producing enzymes into microbes, which then ferment simple sugars to produce structurally identical HMOs. For example, DSM-Firmenich’s 2024 partnership with Lallemand Health Solutions utilized precision fermentation to produce 2’-FL and 3’-SL for synbiotic infant formulas, demonstrating scalability and cost-efficiency. Alternative methods include chemical synthesis and enzymatic catalysis, but precision fermentation is preferred for its precision and sustainability, reducing reliance on dairy-based extraction. Companies like WACKER and BENEO have also adopted this technology, with their 2025 partnership producing 2’-FL for infant formula markets.
Are HMOs a Natural Ingredient?
HMOs are considered natural ingredients when sourced from human breast milk, where they occur abundantly and contribute to infant immunity and gut microbiome health. However, commercially produced HMOs are typically synthetic, created via precision fermentation to replicate the molecular structure of breast milk HMOs. These bio-identical HMOs are chemically indistinguishable from their natural counterparts, ensuring equivalent functionality, such as prebiotic effects and pathogen inhibition. Regulatory bodies, like China’s Ministry of Agriculture and Rural Affairs, recognize these synthetic HMOs as safe and effective, as evidenced by the biosafety approval of Chr. Hansen’s MyOli blend. While not extracted directly from breast milk, their structural fidelity and clinical benefits position them as natural equivalents in premium infant nutrition, meeting consumer demand for breast milk substitutes that closely mimic nature’s design.
Human Milk Oligosaccharides (HMO) Milk Powder Market Overview:
The Human Milk Oligosaccharides (HMO) Milk Powder Market is experiencing robust demand due to the growing approval for the HMO milk powder from regulatory authorities. In August 2024, DSM-Firmenich announced the successful approval of its human milk oligosaccharide, 3-fucosyllactose (3-FL) and lacto-N-fucopentaose I/2'-fucosyllactose (LNFP-I/2'-FL) mixture. This approval is for the novel food ingredients in the United Kingdom and the European Union. This is a significant development, opening up new possibilities for further product innovation and development. Additionally, the increased awareness about HMOs, such as improved gut health and immune support, is fueling market growth. Human milk oligosaccharides (HMOs), a key component of breast milk after lactose and lipids, were approved for use in various infant formula types in China in October 2023. This regulatory change has accelerated market growth, with companies like Novonesis, a leader in HMO development for the U.S. and Europe, now expanding into China. Other notable players include International Flavours and Fragrance (IFF), DSM-Firmenich, and local producer Mengniu. The HMO market is driven by major U.S.-based companies leading innovation in products such as infant formula, dietary supplements, functional foods, and beverages. Rapid urbanization and growing incomes in countries like China, India, and Southeast Asia are driving market expansion. Increased awareness of HMO milk powder benefits, coupled with rising disposable incomes, is fueling demand for premium alternatives to traditional food supplements. Some of the major players covered in this report include BASF SE, Abbott Laboratories, Chr. Hansen Holding A/S, DuPont de Nemours, Inc., DSM-Firmenich, Royal FrieslandCampina N.V., ZuChem, Inc., and Inbiose NV, among others.
Human Milk Oligosaccharides (HMO) Milk Powder Market Trends:
The Human Milk Oligosaccharides (HMO) Milk Powder Market is experiencing dynamic growth, propelled by advancements in HMO production technology and rising demand for premium infant nutrition. A key trend is the adoption of HMO fermentation, particularly through microbial metabolic engineering, which enables scalable production of animal-free HMOs like 2’-FL and LNnT. Companies like Chr. Hansen leverage microbial metabolic engineering to program yeast and bacteria to synthesize HMOs, ensuring bio-identical structures to those in breast milk. Their MyOli 5-HMO blend approval in China highlights this trend’s global impact. Enzymatic synthesis of HMO is another emerging trend, offering precise and sustainable production methods. DSM-Firmenich’s 2024 partnership with Lallemand utilized enzymatic synthesis to produce 2’-FL and 3’-SL for synbiotic formulas, enhancing gut microbiome health. The shift toward animal-free HMO reflects consumer preference for ethical, sustainable ingredients, reducing reliance on dairy-based extraction. WACKER’s 2025 collaboration with BENEO to produce 2’-FL via HMO fermentation further underscores the focus on eco-friendly HMO production technology, driving innovation and accessibility in the pediatric nutrition market.
Human Milk Oligosaccharides (HMO) Milk Powder Market Drivers:
Increasing Demand for Premium Infant Nutrition: The rising consumer preference for premium infant nutrition is a primary driver of the HMO milk powder market, as parents seek formulas that closely replicate the immunological and nutritional benefits of human breast milk. HMOs, such as 2’-Fucosyllactose (2’-FL), support gut microbiome health and infant immunity, making them highly desirable in pediatric nutrition. With growing awareness of early nutrition’s impact on long-term health, particularly in urban areas with high working-mother populations, demand for HMO-enriched breast milk substitutes is surging. For instance, Nestlé’s 2023 launch of Wyeth Illuma with 2’-FL and LNnT in China reflects the market’s focus on delivering scientifically advanced formulas tailored to infant needs. Social media and healthcare professional endorsements further amplify consumer trust in HMO-based products, positioning them as a premium choice for early life nutrition and driving market growth in both developed and emerging regions.
Advancements in HMO Production Technology: Innovations in HMO production technology, particularly HMO fermentation and microbial metabolic engineering, are accelerating market expansion by enabling scalable, cost-effective production of animal-free HMO. Companies like Chr. Hansen utilize genetically engineered microbes to synthesize HMOs like 2’-FL and Lacto-N-neotetraose, replicating the structure of breast milk HMOs with high precision. Their 2024 approval of the MyOli 5-HMO blend in China exemplifies how HMO fermentation supports market growth by meeting regulatory and consumer demands. Enzymatic synthesis HMO methods further enhance production efficiency, as seen in DSM-Firmenich’s 2024 partnership with Lallemand to produce 2’-FL and 3’-SL for synbiotic formulas. These technological advancements reduce reliance on dairy-based extraction, aligning with sustainability goals and expanding the availability of HMOs in premium infant nutrition, thus driving market adoption across global markets.
Growing Focus on Gut Microbiome Health: The increasing recognition of gut microbiome health as a cornerstone of infant immunity and long-term well-being is a significant driver for the HMO milk powder market. HMOs act as prebiotics, fostering beneficial gut bacteria like Bifidobacterium and inhibiting pathogens, which reduces infection risks and supports digestive health. A 2024 study highlighted that HMO-supplemented formulas enhance microbiome profiles in formula-fed infants, closely mimicking those of breastfed infants. This scientific validation fuels demand for HMO-enriched breast milk substitutes, particularly among health-conscious parents and pediatricians. The trend extends beyond infants, with HMOs being explored for adult nutrition, broadening market potential. As consumer education on the gut-immune axis grows, driven by healthcare campaigns and digital platforms, the early life nutrition market increasingly prioritizes HMO-based products, cementing their role in pediatric nutrition.
Human Milk Oligosaccharides (HMO) Milk Powder Market Restraints:
High Costs of HMO Production Technology: The HMO milk powder market faces a significant restraint due to the high costs associated with HMO production technology, particularly HMO fermentation and enzymatic synthesis of HMO. Producing animal-free HMO requires advanced biotechnological processes, including microbial metabolic engineering, which involve substantial investments in research, development, and specialized facilities. For example, WACKER’s 2025 partnership with BENEO to produce 2’-FL highlighted the complexity and cost of scaling HMO fermentation, which increases product prices and limits affordability in price-sensitive markets. These high costs pose a barrier to widespread adoption, particularly in developing regions where budget constraints drive preference for conventional formulas. While advancements in HMO production technology are improving scalability, current cost structures hinder market penetration, requiring ongoing innovation to reduce expenses and enhance accessibility of HMO-enriched premium infant nutrition.
Complex Regulatory Landscape for Pediatric Nutrition: The HMO milk powder market is constrained by a complex and stringent regulatory landscape governing pediatric nutrition products. HMOs, as novel food ingredients, require rigorous safety and efficacy evaluations before market approval, which vary significantly across regions. For instance, China’s 2023 approval of 2’-FL and LNnT as food nutrition enhancers involved extensive biosafety assessments, delaying product launches. These regulatory hurdles increase compliance costs and timelines, particularly for animal-free HMO produced via HMO fermentation. Inconsistent global standards further complicate market entry, as manufacturers must navigate diverse requirements for breast milk substitutes. This restraint limits the speed of innovation and market expansion, particularly for smaller companies lacking resources to meet regulatory demands, slowing the adoption of HMO-enriched products in the early life nutrition market.
Human Milk Oligosaccharides (HMO) Milk Powder Market Segmentation Analysis:
By Type, 2'-Fucosyllactose (2'FL) is rising significantly: Among the types of HMOs, 2'-Fucosyllactose (2'FL) dominates due to its prevalence in human breast milk and well-documented benefits for gut microbiome health and infant immunity. As the most abundant HMO in breast milk, 2'FL acts as a prebiotic, promoting Bifidobacterium growth and inhibiting pathogen adhesion in the infant gut. Its widespread adoption in premium infant nutrition stems from its proven ability to enhance digestive health and reduce infection risks. For instance, Nestlé’s 2023 launch of Wyeth Illuma infant formula enriched with 2'FL in China underscores its market leadership, driven by consumer demand for breast milk substitutes that mimic natural benefits. Advances in HMO fermentation and enzymatic synthesis have made 2'FL production scalable, with companies like Chr. Hansen expanding its use in pediatric nutrition globally. Its versatility also extends to functional foods, solidifying 2'FL as the leading HMO type.
Infant Formula is the most popular application: Infant Formula is the primary application for HMO milk powders, driven by the global demand for breast milk substitutes that replicate the immunological and nutritional benefits of human milk. HMOs like 2'FL and LNnT enhance infant formula by supporting gut microbiome health and infant immunity, addressing the needs of infants who cannot be breastfed. The application’s dominance is fueled by parental awareness of early nutrition’s impact on long-term health, particularly in reducing infections and supporting cognitive development. For example, Abbott’s 2024 Similac formula with a five-HMO blend, including 2'FL and 3’-SL, highlights the trend toward premium infant nutrition that closely mimics breast milk. Regulatory approvals, such as China’s 2023 authorization of HMOs in infant formula, further boost this segment’s growth, making infant formula the cornerstone of the early life nutrition market.
Asia Pacific is expected to lead the market expansion: The Asia Pacific region leads the HMO milk powder market due to its large infant population, rising disposable incomes, and increasing awareness of pediatric nutrition. Countries like China and India drive demand for HMO-enriched infant formula, fueled by high working-mother populations and lower breastfeeding rates in urban areas. The region’s focus on premium infant nutrition is evident in Nestlé’s 2023 launch of HMO-fortified Wyeth Illuma in China, targeting health-conscious parents. Additionally, regulatory advancements, such as China’s 2024 biosafety approval for Chr. Hansen’s MyOli 5-HMO blend facilitates market expansion by enabling broader HMO integration. The Asia Pacific region’s robust HMO production technology infrastructure, including partnerships like WACKER and BENEO’s 2025 2'FL production, further strengthens its position as the dominant market for HMO milk powders.
Human Milk Oligosaccharides (HMO) Milk Powder Market Key Developments:
In February 2025, a2 Milk Company launched its new a2 Genesis infant formula specifically for the Chinese market. This premium product is notable for its inclusion of a three-HMO blend, featuring 2?-fucosyllactose (2?-FL), 3?-sialyllactose (3?-SL), and 6?-sialyllactose (6?-SL). The formula also incorporates algal DHA, prebiotic GOS, and the probiotic strain Bifidobacterium lactis HN019, reflecting the market trend toward multi-functional, comprehensive infant nutrition that goes beyond a single HMO to provide more holistic benefits. This launch capitalizes on the growing consumer demand in China for high-end, scientifically-backed infant formulas.
In March 2024, Chr. Hansen, which merged with Novozymes to form Novonesis, successfully passed the biosafety review by China's Ministry of Agriculture and Rural Affairs for all five single HMOs in its MyOli blend. This milestone positioned the company to be a major player in the expanding Chinese infant formula market. The approval of a comprehensive blend, including 3'-fucosyllactose (3'-FL), is particularly noteworthy as it aligns with the empirical studies on the composition of breast milk from Chinese mothers, making the product highly relevant for the local market.
In January 2024, FrieslandCampina Ingredients announced it had received regulatory approval from the Thai Food and Drug Administration to use its 2'-fucosyllactose (2'-FL) HMO in infant and follow-up formulas. This approval was a significant step for the company, as it made them the exclusive supplier of this specific HMO component in Thailand at the time. This development not only opened up a new market for FrieslandCampina but also provided opportunities for infant formula manufacturers in the region to develop new products with this key ingredient.
List of Top Human Milk Oligosaccharides (HMO) Milk Powder Companies:
Nestlé S.A.
Abbott Laboratories
Chr. Hansen Holding A/S
DSM-Firmenich AG
BENEO GmbH
Human Milk Oligosaccharides (HMO) Milk Powder Market Scope:
| Report Metric | Details |
|---|---|
| Total Market Size in 2025 | USD 3.2 billion |
| Total Market Size in 2030 | USD 5.4 billion |
| Forecast Unit | Billion |
| Growth Rate | 11.2% |
| Study Period | 2020 to 2030 |
| Historical Data | 2020 to 2023 |
| Base Year | 2024 |
| Forecast Period | 2025 – 2030 |
| Segmentation | Type, Application, Distribution Channel, Geography |
| Geographical Segmentation | North America, South America, Europe, Middle East and Africa, Asia Pacific |
| Companies |
|
Human Milk Oligosaccharides (HMO) Milk Powder Market Segmentation:
By Type
2'-Fucosyllactose (2'FL)
3'-Fucosyllactose (3'FL)
3'-Sialyllactose (3'SL)
6'-Sialyllactose (6'SL)
Lacto-N-tetraose (LNT)
Lacto-N-neotetraose (LNnT)
Difucosyllactose (DFL)
By Application
Infant Formula
Dietary Supplements
Functional Foods and Beverages
Cosmetics
By Distribution Channel
Online Retailers
Supermarkets/hypermarkets
Specialty Stores
Convenience Stores
By Region
North America
Europe
Asia Pacific
South America
Middle East & Africa
Market Segmentation
By Type
By Distribution Channel
By Application
By Geography
Table of Contents
1. EXECUTIVE SUMMARY
2. MARKET SNAPSHOT
2.1. Market Overview
2.2. Market Definition
2.3. Scope of the Study
2.4. Market Segmentation
3. BUSINESS LANDSCAPE
3.1. Market Drivers
3.2. Market Restraints
3.3. Market Opportunities
3.4. Porter’s Five Forces Analysis
3.5. Industry Value Chain Analysis
3.6. Policies and Regulations
3.7. Strategic Recommendations
4. TECHNOLOGICAL OUTLOOK
5. HUMAN MILK OLIGOSACCHARIDES (HMO) MILK POWDER MARKET BY TYPE
5.1. Introduction
5.2. 2'-Fucosyllactose (2'FL)
5.3. 3'-Fucosyllactose (3'FL)
5.4. 3'-Sialyllactose (3'SL)
5.5. 6'-Sialyllactose (6'SL)
5.6. Lacto-N-tetraose (LNT)
5.7. Lacto-N-neotetraose (LNnT)
5.8. Difucosyllactose (DFL)
6. HUMAN MILK OLIGOSACCHARIDES (HMO) MILK POWDER MARKET BY DISTRIBUTION CHANNEL
6.1. Introduction
6.2. Online Retailers
6.3. Supermarket/ Hypermarket
6.4. Specialty Stores
6.5. Convinience Stores
7. HUMAN MILK OLIGOSACCHARIDES (HMO) MILK POWDER MARKET BY APPLICATION
7.1. Introduction
7.2. Infant Nutrition
7.3. Dietery Supplements
7.4. Functional Foods and Beverages
7.5. Cosmetics
8. HUMAN MILK OLIGOSACCHARIDES (HMO) MILK POWDER MARKET BY GEOGRAPHY
8.1. Introduction
8.2. North America
8.2.1. USA
8.2.2. Canada
8.2.3. Mexico
8.3. South America
8.3.1. Brazil
8.3.2. Argentina
8.3.3. Others
8.4. Europe
8.4.1. Germany
8.4.2. France
8.4.3. United Kingdom
8.4.4. Spain
8.4.5. Others
8.5. Middle East and Africa
8.5.1. Saudi Arabia
8.5.2. UAE
8.5.3. Others
8.6. Asia Pacific
8.6.1. China
8.6.2. Japan
8.6.3. India
8.6.4. South Korea
8.6.5. Thailand
8.6.6. Indonesia
8.6.7. Others
9. COMPETITIVE ENVIRONMENT AND ANALYSIS
9.1. Major Players and Strategy Analysis
9.2. Market Share Analysis
9.3. Mergers, Acquisitions, Agreements, and Collaborations
9.4. Competitive Dashboard
10. COMPANY PROFILES
10.1. Nestlé S.A.
10.2. Abbott
10.3. Novonesis
10.4. DSM-Firmenich
10.5. BENEO
10.6. Wacker Chemie AG
10.7. BASF SE
10.8. International Flavors & Fragrances Inc
10.9. FrieslandCampina
10.10. Biosynth
11. APPENDIX
11.1. Currency
11.2. Assumptions
11.3. Base and Forecast Years Timeline
11.4. Key benefits for the stakeholders
11.5. Research Methodology
11.6. Abbreviations
LIST OF FIGURES
LIST OF TABLES
Request Customization
Tell us your specific requirements and we will customize this report for you.
Download Free Sample
Get a sample copy of this report with charts, TOC, and methodology.
Speak to Analyst
Ask our analysts any questions you have about this market research report.
Human Milk Oligosaccharides (HMO) Milk Powder Market Report
Trusted by the world's leading organizations











